Ni 200 is a commercially pure wrought nickel alloy known for its exceptionally high nickel content (typically ≥99%) and excellent corrosion resistance in a wide range of environments. Due to its purity, Ni 200 exhibits outstanding electrical and thermal conductivity, making it widely used in electronics, chemical processing, and alkaline applications. Understanding the chemical composition of Ni 200 is essential for evaluating its performance in corrosive and high-temperature environments.
Introduction to Ni 200 Material
Ni 200 (UNS N02200) is a solid-solution strengthened nickel alloy with very low impurity levels. Its high purity gives it superior resistance to caustic environments and excellent mechanical properties in both hot and cold working conditions.
Ni 200 Chemical Composition Overview
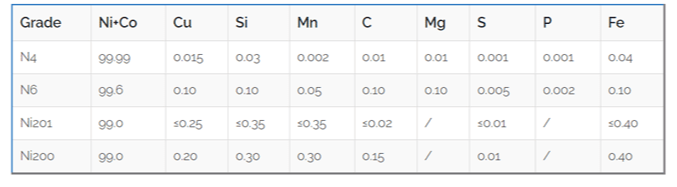
| Element | Content (%) |
|---|---|
| Nickel (Ni) | ≥ 99.0 |
| Iron (Fe) | ≤ 0.40 |
| Copper (Cu) | ≤ 0.25 |
| Manganese (Mn) | ≤ 0.35 |
| Carbon (C) | ≤ 0.15 |
| Silicon (Si) | ≤ 0.35 |
| Sulfur (S) | ≤ 0.01 |
High Nickel Content (≥99%) and Its Role
The extremely high nickel content provides Ni 200 with excellent corrosion resistance, particularly in alkaline environments, as well as high ductility and toughness. It also contributes to superior electrical and thermal conductivity.
Iron (Fe) Content and Its Effect
Iron is kept at low levels to maintain corrosion resistance and purity. Excessive iron can reduce resistance to chemical attack and slightly decrease conductivity.
Effect of Carbon (C) Content on Properties
Carbon content is carefully controlled in Ni 200. Higher carbon levels can lead to graphite formation at elevated temperatures, which may reduce mechanical properties and ductility.
Role of Manganese (Mn) and Silicon (Si)
| Element | Function |
|---|---|
| Manganese (Mn) | Improves hot workability and helps control sulfur |
| Silicon (Si) | Acts as a deoxidizer during melting |
Control of Copper (Cu) and Trace Elements
Copper and other trace elements are kept at low levels to maintain the alloy’s purity and ensure consistent corrosion resistance and conductivity.
Sulfur (S) Content and Machinability
Sulfur is strictly limited because it can cause hot shortness and reduce ductility. While sulfur can improve machinability in some alloys, in Ni 200 it is minimized to preserve mechanical integrity.
Effect of Composition on Electrical Conductivity
The high purity of Ni 200 results in excellent electrical conductivity compared to alloyed nickel materials. Impurities such as iron and carbon can reduce conductivity, which is why they are tightly controlled.
Effect of Composition on Corrosion Resistance
Ni 200 exhibits excellent corrosion resistance in neutral and alkaline environments. Its high nickel content makes it particularly resistant to caustic solutions and organic chemicals.
Performance in Alkaline Environments (e.g., Caustic Soda)
Ni 200 is widely used in caustic soda (NaOH) systems due to its outstanding resistance to alkaline corrosion. It maintains stability even at elevated temperatures in concentrated alkaline solutions.
Role of High-Purity Nickel in High-Temperature Performance
Ni 200 performs well at moderate high temperatures, but at temperatures above approximately 315°C, carbon can precipitate as graphite. For higher temperature applications, Ni 201 (with lower carbon content) is preferred.
Difference Between Ni 200 and Ni 201 Composition
| Property | Ni 200 | Ni 201 |
|---|---|---|
| Carbon Content | ≤ 0.15% | ≤ 0.02% |
| High-Temperature Stability | Moderate | Better |
| Graphitization Risk | Higher | Lower |
Standards and Material Designations
| Standard | Designation |
|---|---|
| UNS | N02200 |
| ASTM | B160 |
Effect of Composition on Typical Applications
- Electronics: due to high conductivity and purity
- Chemical processing: resistance to caustic environments
- Food processing: corrosion resistance and cleanliness
- Heat exchangers: good thermal conductivity
Frequently Asked Questions (FAQ)
What is the nickel content of Ni 200?
Ni 200 contains at least 99% nickel, making it a commercially pure nickel alloy.
Is Ni 200 suitable for high-temperature use?
It is suitable for moderate temperatures, but for higher temperatures Ni 201 is preferred due to lower carbon content.
Why is Ni 200 used in alkaline environments?
Because of its excellent resistance to caustic corrosion, especially in sodium hydroxide systems.
What is the main difference between Ni 200 and Ni 201?
The primary difference is carbon content, with Ni 201 having much lower carbon for better high-temperature stability.



